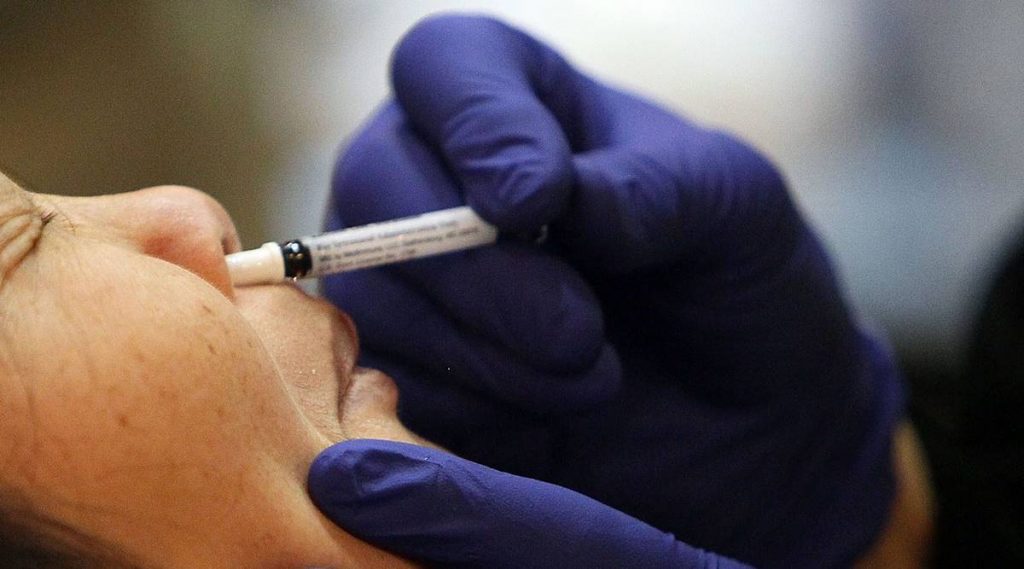
New Delhi: The Drugs Controller General of India (DCGI) approved Tuesday Bharat Biotech’s intranasal Covid-19 vaccine for restricted emergency use in those aged above 18 years. “Big Boost to India’s Fight against Covid-19! Bharat Biotech’s ChAd36-SARS-CoV-S Covid-19 (Chimpanzee Adenovirus Vectored) recombinant nasal vaccine approved by @CDSCO_INDIA_INF for primary immunization against Covid-19 in 18+ age group for restricted use in emergency situation,” Union Health Minister Mansukh Mandaviya tweeted.
Mandaviya said this step will further strengthen ‘our collective fight’ against the pandemic. India has harnessed its science, research and development (R&D), and human resources in the fight against Covid-19 under Prime Minister Narendra Modi’s leadership, he added.
“With the science-driven approach & Sabka Prayas (efforts of everyone), we will defeat Covid-19,” Mandaviya asserted.
Hyderabad-based Bharat Biotech International Limited (BBIL) completed clinical trials of the nasal vaccine with about 4,000 volunteers and there is no side effect or adverse reaction reported so far, company sources said.
In August, Bharat Biotech had said that its Covid-19 intranasal vaccine (BBV154) has proven to be safe, well-tolerated, and immunogenic in subjects in controlled clinical trials phase -3.
Also Read – IT raids at Rajasthan minister Rajendra Yadav’s premises
BBV154 has been specifically formulated to allow intranasal delivery. In addition, the nasal delivery system has been designed and developed to be cost-effective in low and middle-income countries, the vaccine maker had said.
“Being an intranasal vaccine, BBV154 may produce local antibodies in the upper respiratory tract. These may provide the potential to reduce infection and transmission. Further studies are being planned,” Bharat Biotech had earlier said.
Two separate and simultaneous clinical trials were conducted to evaluate BBV154 as a primary dose (2-dose) schedule and a heterologous booster dose for subjects who have previously received 2 doses of the two commonly administered Covid vaccines – Covishield and Covaxin – in India.
Separately, the DCGI also granted permission to the firm to conduct phase-3 clinical trial to compare the immunogenecity and safety of BBV154 (intranasal) with Covaxin. This trial was conducted at nine sites.